A three-year regulatory grind to bring Bedrocan to Ireland.
Bedrocan is the only producer of standardised, pharmaceutical-grade cannabis flos that meets European pharmacopoeia standards for medicinal use.

Oleo Technologies supplies pharmaceutical-grade Bedrocan cannabis-based medicinal products to MCAP-registered prescribers and Section 56 dispensing pharmacies across Ireland. Founded 2019. Bedrocan import licence granted 2021. CRO 651282.
Access the prescriber portal for product information, batch availability, Certificates of Analysis, and direct supply ordering. Verification required.
Enter prescriber portal Patient or family memberRead about the Medical Cannabis Access Programme, eligible conditions under the Misuse of Drugs (Amendment) Regulations 2017, and the prescribing pathway via your healthcare professional.
Read about MCAPThe Medical Cannabis Access Programme was established under the Misuse of Drugs (Amendment) Regulations 2017. Eligibility is condition-specific and statutorily defined. Cannabis-based medicinal products under MCAP can only be prescribed by a consultant for a patient who has not responded to standard treatments.
Speak to your healthcare professional. The prescribing pathway begins with a consultant referral; Oleo supplies the medicine through Section 56 dispensing pharmacies once a valid private prescription is in place.
Misuse of Drugs (Amendment) Regulations 2017 (S.I. No. 262 of 2017). Programme administered by the Health Products Regulatory Authority. Eligibility determined by the prescribing consultant.
Bedrocan is the only producer of standardised, pharmaceutical-grade cannabis flos that meets European pharmacopoeia standards for medicinal use.

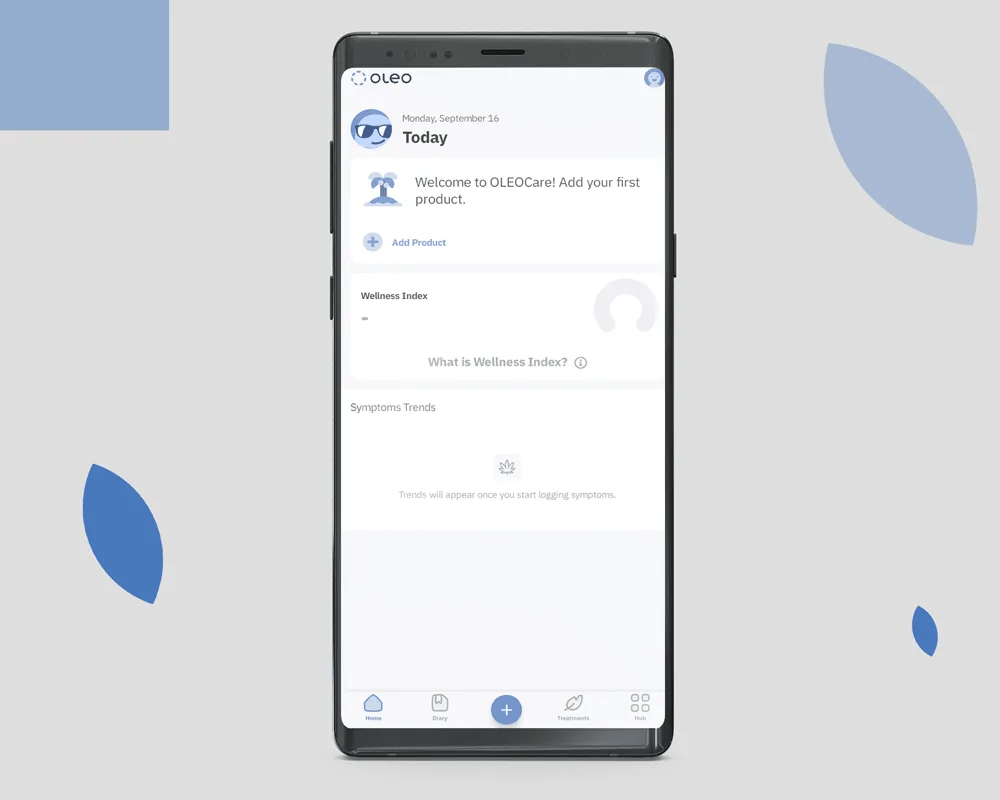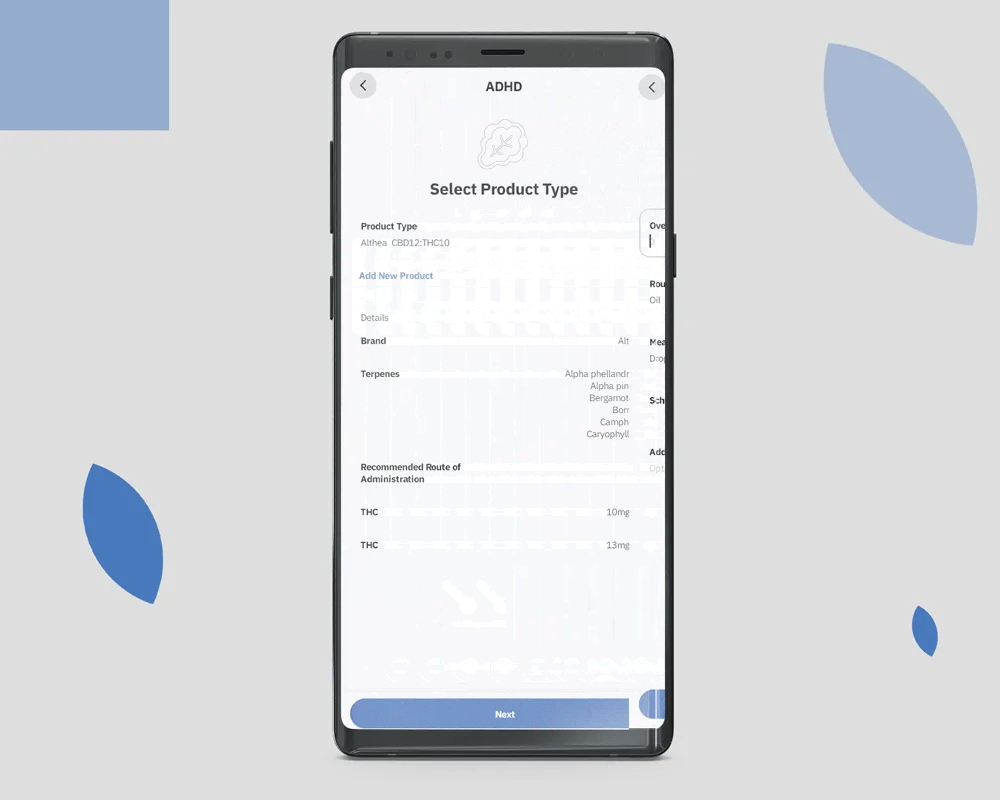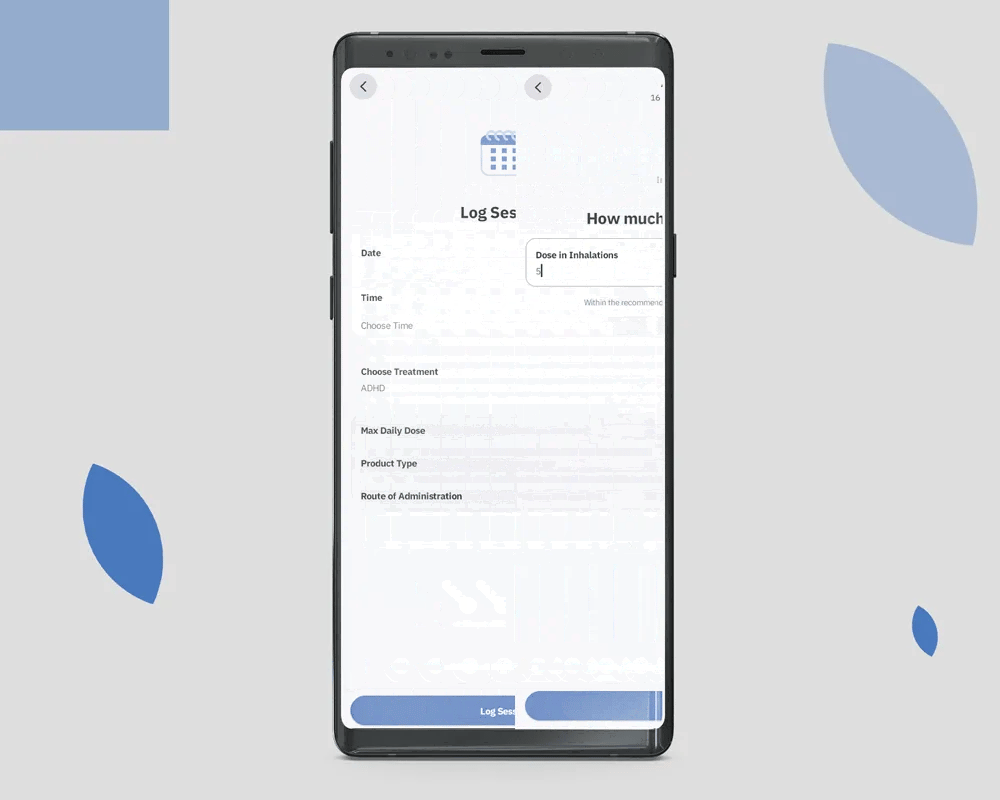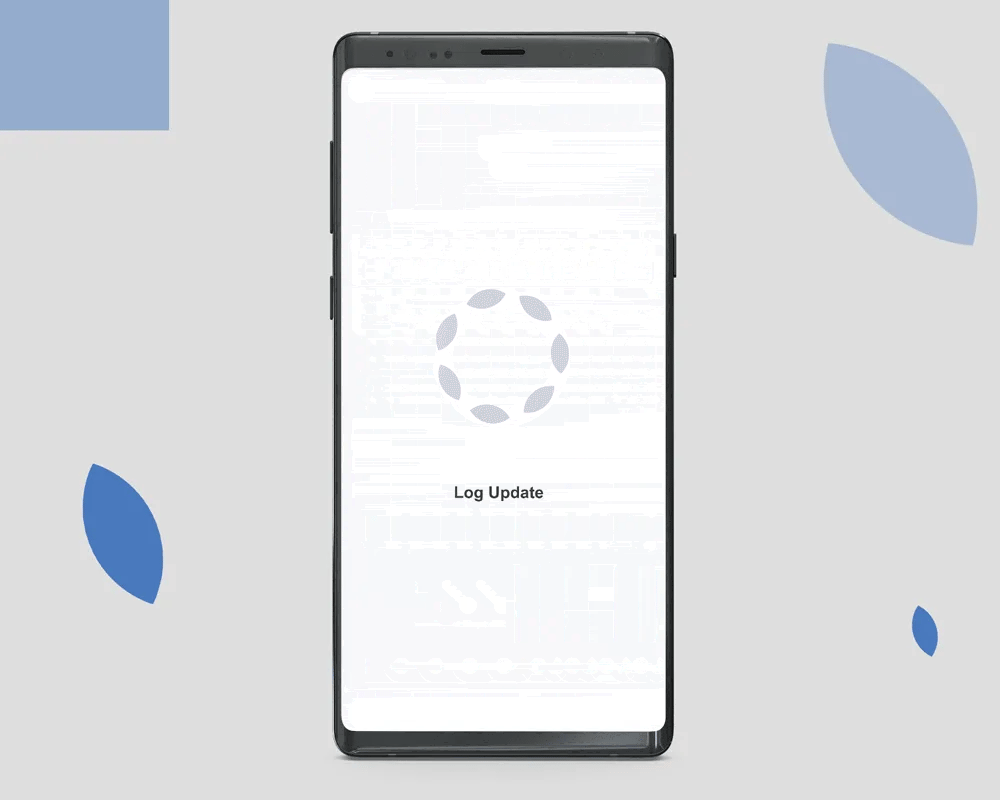
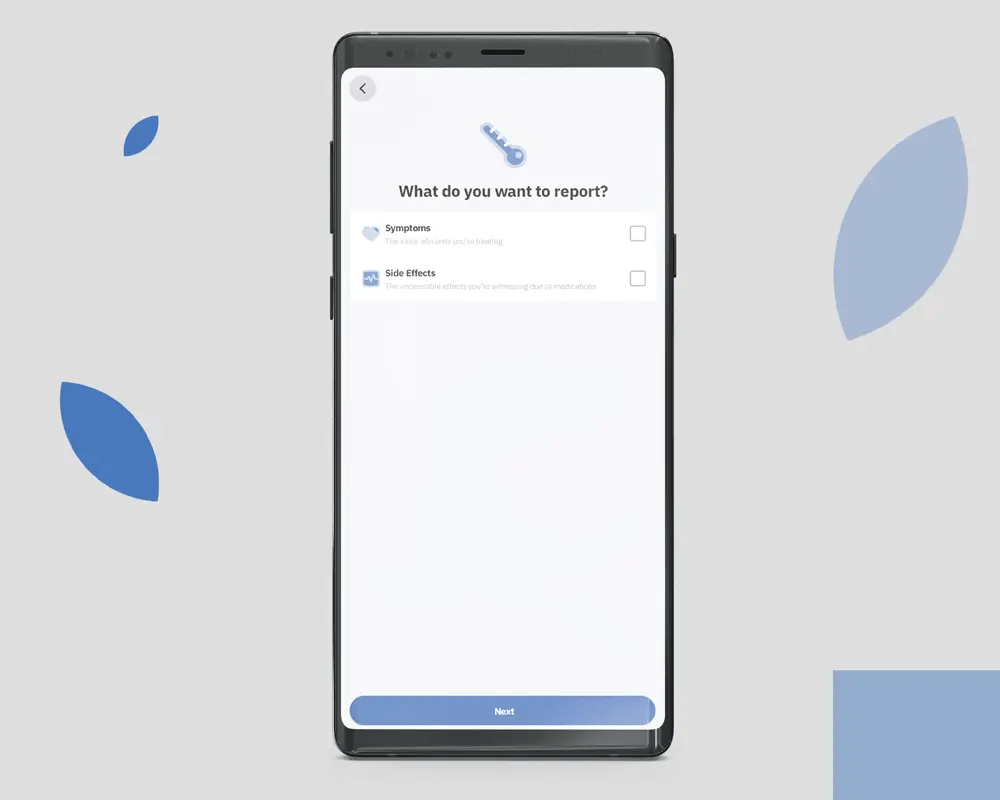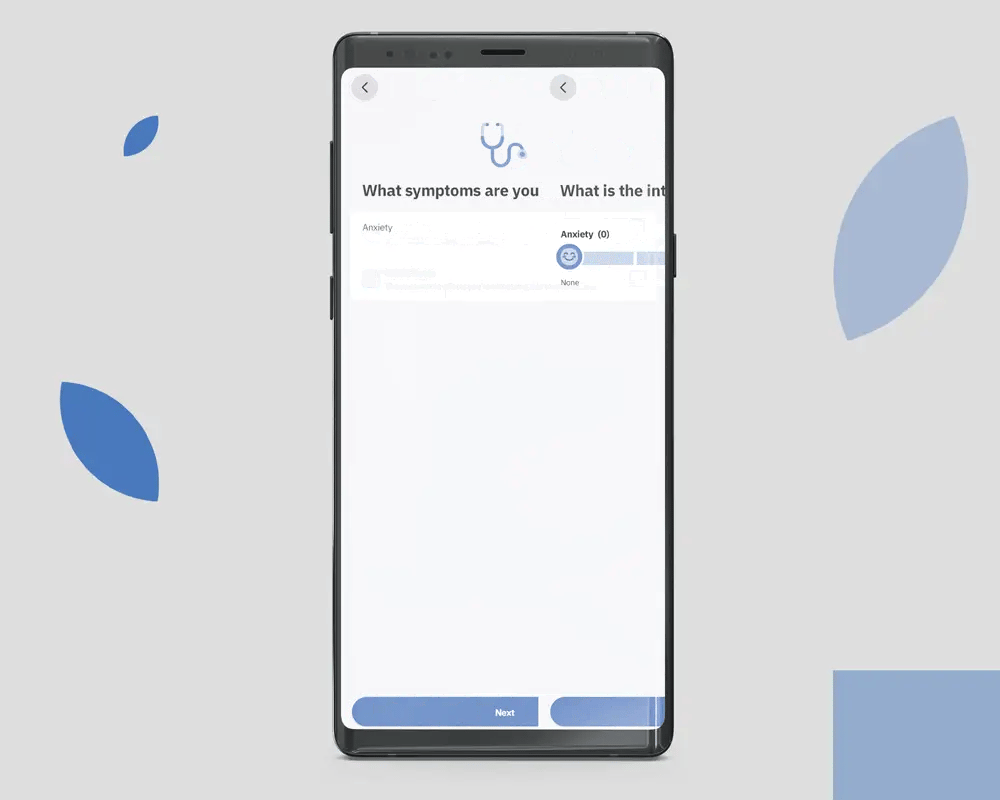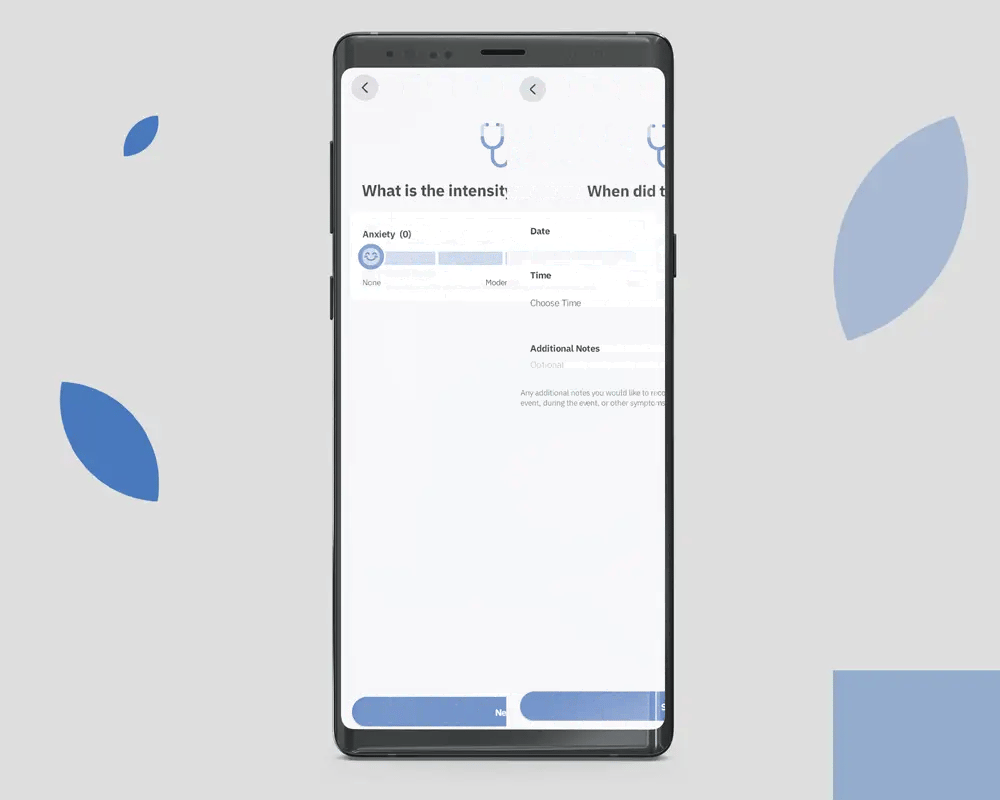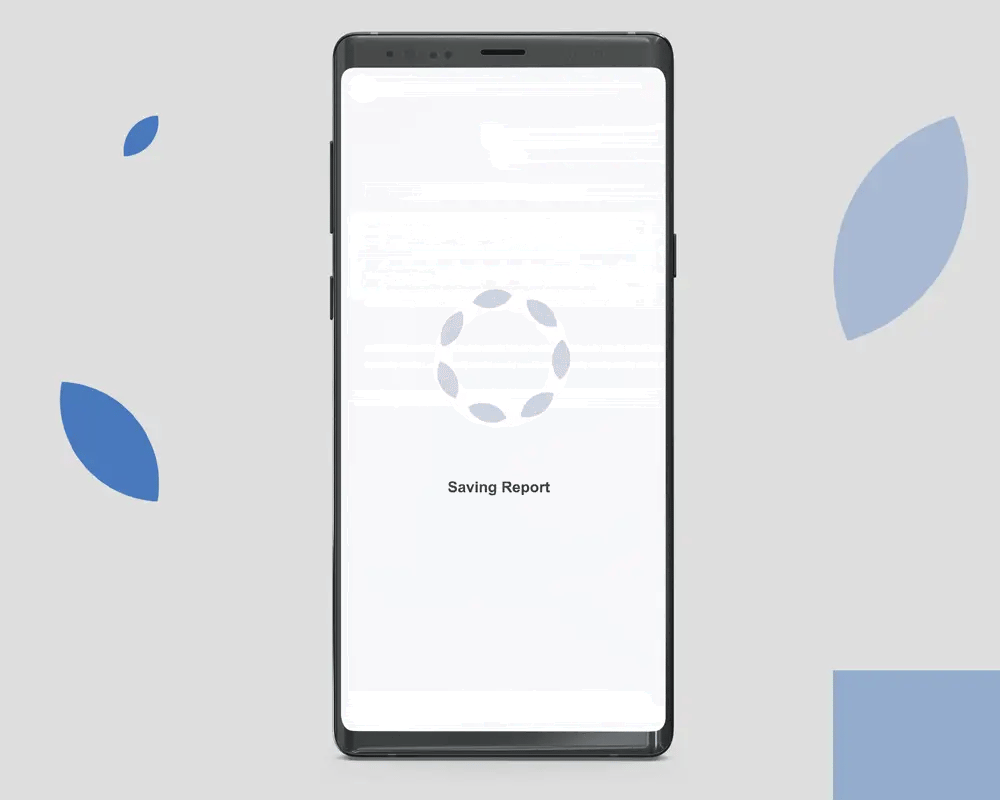
OleoCare is the patient app for those already enrolled in MCAP and on a valid prescription. It is not a marketing app and is not available to the general public. Patients access it through their prescriber after enrolment.
Patients log symptoms on a 0 to 10 scale across the conditions their prescriber is monitoring. The data is theirs. It is shared with the prescribing clinician at the patient's discretion.
Reminders are configured by the prescriber, not the patient. Adjusting dose without prescriber consultation is not supported in the app.
Special-category health data handled under Article 9 of GDPR. Data Protection Impact Assessment on file. No third-party tracking or advertising SDKs.
Prescribing clinicians see longitudinal symptom data for the patients they have added. The clinical relationship is preserved; the supplier is the supply backbone.





The Panacea vaporiser is the delivery device commissioned by Oleo for prescribed MCAP patients. Convection-based heating, controlled temperature ranges, no combustion. Designed to align with the European Pharmacopoeia 2.9.18 inhalation monograph.

The job here is to be a quietly dependable backbone. Patients have a clinical relationship with their prescriber. Our role is to make sure the medicine is on the pharmacy shelf when the prescription arrives.
Oleo Technologies was co-founded in February 2019 by Richard Creagh and Shirley Creagh. Richard spent 25 years in medtech sales, including five years at Applied Medical, where he was promoted to National Sales Manager and launched the Advanced Energy Devices line. Shirley is co-founder and Chief Technology Officer; her background spans technology operations and regulated systems.
The Bedrocan import licence took three years of regulatory and compliance work to land. That sequence, the medtech background and the regulatory grind, is the spine of how Oleo operates today.
Stated in plain regulatory English, sourced from the legislation. No marketing language.
S.I. No. 262 of 2017. Establishes the Medical Cannabis Access Programme, the eligible conditions, the prescribing requirements, and the supply chain for cannabis-based medicinal products in Ireland.
Oleo is an HPRA-listed MCAP supplier since November 2021. Annual licence renewal, batch documentation, and pharmacovigilance reporting under HPRA prescribing guidance.
The 7th edition Code prohibits direct-to-public advertising of prescription-only medicines. Product-specific information sits behind the prescriber portal gate. Public pages describe the company and the programme only.
Good Manufacturing Practice (GMP) supply requirements. Bedrocan is supplied to Oleo under European GMP; documentation accompanies every batch.
Storage, dispensing, record-keeping, and disposal at pharmacy level. CDR/3 register required at every Section 56 dispensing pharmacy handling Bedrocan.
OleoCare patient app handles Article 9 special-category data. Data Protection Impact Assessment on file with the Data Protection Commission. No third-party tracking SDKs.
One-page MCAP enrolment summary, current product list with batch availability, and a direct line to clinical and regulatory support. Verification required.
Enter the prescriber portalThe prescribing pathway begins with a consultant referral. We do not provide medical advice and do not communicate with patients outside the OleoCare app, which is enrolled-patients-only.
Read the patient guide to MCAP